|
Related Topics: |
|
|
|
Current News |
|
Chemistry A to Z |
|
About Internetchemistry |
Microfluidic chambers advance the science of growing neurons |
|
The technique is described in the journal of the Royal Society of Chemistry � Lab on a Chip. �This finding will be very positively greeted by the neuroscience community,� said Martha Gillette, who is an author on the study and the head of the cell and developmental biology department at Illinois. �This is pushing the limits of what you can do with neurons in culture.� |
|
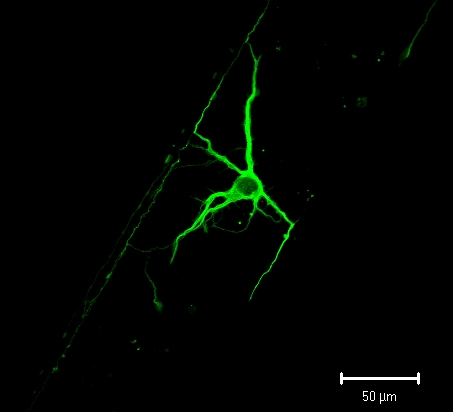
Growing viable mammalian neurons at low density in an artificial environment is no easy task. Using postnatal neurons only adds to the challenge, Gillette said, because these cells are extremely sensitive to environmental conditions. All neurons rely on a steady supply of proteins and other �trophic factors� present in the extracellular fluid. These factors are secreted by the neurons themselves or by support cells, such as the glia. This is why neurons tend to do best when grown at high density and in the presence of other brain cells. But a dense or complex mixture of cells complicates the task of characterizing the behavior of individual neurons. One technique for keeping neural cultures alive is to grow the cells in a medium that contains serum, or blood plasma. This increases the viability of cells grown at low density, but it also �contaminates� the culture, making it difficult to determine which substances were produced by the cells and which came from the serum. Those hoping to understand the cellular origins of trophic factors in the brain would benefit from a technique that allows them to measure the chemical outputs of individual cells. The research team made progress toward this goal by addressing a few key obstacles. First, the researchers scaled down the size of the fluid-filled chambers used to hold the cells. Chemistry graduate student Matthew Stewart made the small chambers out of a molded gel of polydimethylsiloxane (PDMS). The reduced chamber size also reduced � by several orders of magnitude � the amount of fluid around the cells, said Biotechnology Center director Jonathan Sweedler, an author on the study. This �miniaturization of experimental architectures� will make it easier to identify and measure the substances released by the cells, because these �releasates� are less dilute. �If you bring the walls in and you make an environment that�s cell-sized, the channels now are such that you�re constraining the releasates to physiological concentrations, even at the level of a single cell,� Sweedler said. Second, the researchers increased the purity of the material used to form the chambers. Cell and developmental biology graduate student Larry Millet exposed the PDMS to a series of chemical baths to extract impurities that were killing the cells. Millet also developed a method for gradually perfusing the neurons with serum-free media, a technique that resupplies depleted nutrients and removes cellular waste products. The perfusion technique also allows the researchers to collect and analyze other cellular secretions � a key to identifying the biochemical contributions of individual cells. �We know there are factors that are communicated in the media between the cells,� Millet said. �The question is what are they, and how can we get at those?� This combination of techniques enabled the research team to grow postnatal primary hippocampal neurons from rats for up to 11 days at extremely low densities. Prior to this work, cultured neurons in closed-channel devices made of untreated, native PDMS remained viable for two days at best. The cultured neurons also developed more axons and dendrites, the neural tendrils that communicate with other cells, than those grown at low densities with conventional techniques, Gillette said. �Not only have we increased the cells� viability, we�ve also increased their ability to differentiate into what looks much more like a mature neuron,� she said. Sweedler noted that the team�s successes are the result of a unique collaboration among scientists with very different backgrounds. �(Materials science and engineering professor) Ralph Nuzzo is one of the pioneers in self-assembled monolayers and surface chemistry,� Sweedler said. �Martha Gillette�s expertise is in understanding how these neurons grow, and in imaging them. My lab does measurement science on a very small scale. It�s almost impossible for any one lab to do all that.� Nuzzo and Sweedler are William H. and Janet Lycan professors of chemistry. Gillette is Alumni Professor of Cell and Developmental Biology. All are appointed in the Institute for Genomic Biology. Sweedler and Gillette are affiliates of the Beckman Institute and the Neuroscience Program. Sweedler is a professor in the Bioengineering Program and Gillette in the College of Medicine. |
|
|
|