|
Related Topics: |
|
Current News |
|
Chemistry A to Z |
|
About Internetchemistry |
|
- Imprint |
|
|
Berkeley Researchers Identify Photosynthetic Dimmer Switch |
|
The pigment-binding protein CP29, one of the “minor” light-harvesting proteins in green plants, has been identified as a valve that permits or blocks the critical release of excess solar energy during photosynthesis. Furthermore, it has been proposed that the opening and closing of this valve can be controlled by raising or lowering ambient pH levels. |
|
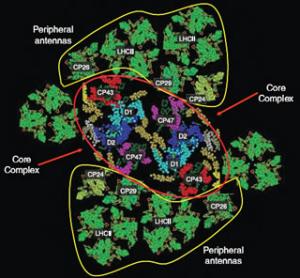
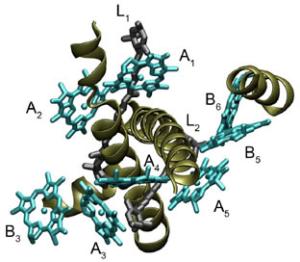
Graham Fleming, a physical chemist who holds joint appointments with the U.S. Department of Energy’s Lawrence Berkeley National Laboratory (Berkeley Lab) and the University of California (UC) at Berkeley, was one the leaders of this study, along with Krishna Niyogi, who also holds a joint appointment with Berkeley Lab and UC Berkeley, and Roberto Bassi, of the University of Verona, Italy. “This is really the first detailed picture ever obtained of the molecular mechanism behind the regulation of light harvesting energy,” Fleming said. “We believe we will soon be in position to build a complete model of the flow of energy through the photosynthetic light harvesting system that will include how the flow is controlled. This model could then be applied to the engineering of artificial versions of photosynthesis.” The results of this study are reported in the journal Science (May 9, 2008) in a paper entitled: “Architecture of a Charge-Transfer State Regulating Light Harvesting in a Plant Antenna Protein.” Co-authoring the paper with Fleming, Niyogi and Bassi were Tae Kyu Ahn, Thomas Avenson, Matteo Ballottari and Yuan-Chung Cheng. Through photosynthesis, green plants are able to harvest energy from sunlight and convert it to chemical energy at an energy transfer efficiency rate of approximately 97 percent. If scientists can create artificial versions of photosynthesis, the dream of solar power as the ultimate green and renewable source of electrical energy could be realized. However, a potential pitfall for any sunlight-harvesting system is that if the system becomes overloaded with absorbed solar energy, it most likely will suffer some form of damage. Plants solve this problem on a daily basis with a photo-protective mechanism called energy-quenching. Excess energy, detected by changes in pH levels, is safely dissipated from one molecular system to another, where it can then be routed down relatively harmless chemical reaction pathways. As Fleming once explained, “This defense mechanism is so sensitive to changing light conditions, it will even respond to the passing of clouds overhead.” In 2005, Fleming and his research group identified zeaxanthin, a member of the carotenoid family of pigment molecules, as the safety outlet in the photo-protection of green plants. A plant’s light harvesting system consists of two protein complexes, Photosystem I and Photosystem II. Each complex features antennae made up of chlorophyll and carotenoid molecules that gain extra “excitation” energy when they capture photons. Fleming and his group found that intense exposure to light triggers the formation of zeaxanthin molecules in Photosystem II. These zeaxanthin molecules interact with excited chlorophyll molecules and dissipate the excess energy via a charge-transfer mechanism - zeaxanthin gives up an electron to the chlorophyll, bringing the chlorophyll’s energy back down to its ground state and turning the zeaxanthin into a radical cation which, unlike an excited chlorophyll molecule, is a non-oxidizing agent. However, until now a critical piece to the puzzle was missing - they did not know how the chlorophyll and zeaxanthin interaction was being regulated. “If this were the murder mystery board game Clue, you could say that we had found the weapon (zeaxanthin) but didn’t know how and precisely where the crime took place,” Fleming said. Fleming and his colleagues knew where to look, however. Recently they’d used near-infrared absorption spectroscopy to demonstrate that the generation of the zeaxanthin radical cation occurs exclusively in the three minor light-harvesting proteins, C29, CP26 and CP24. To determine whether one or all of the minor complexes were responsible for production of the energy-quenching cation, they expressed CP29, CP26 and CP24 in bacteria and reconstituted them in vitro with chlorophyll, zeaxanthin and other Photosystem II proteins. They then used ultrafast pump-probe spectroscopy to follow the energy trail on the femtosecond timescale (a femtosecond is one millionth of a billionth of a second) of the energy-quenching process. “Our findings showed that energy-quenching occurs within all three minor complexes, which is consistent with the results of previous genetic and spectroscopic analyses that indicated no single antenna protein is specifically required for quenching to take place,” said Fleming. To learn more about how the energy quenching process works, Fleming and his colleagues focused their efforts on C29 because the genetics and molecular architecture of this protein have been well characterized and the supply of known mutations is ample. Their findings suggest that the change in pH as a result of excess solar energy results in CP29 undergoing a conformational change which alters the reduction potential of excitonically-coupled chlorophyll molecules. This then promotes the charge-transfer with zeaxanthin that produces radical cations. Furthermore, it appears that this conformational change is reversible, which opens the possibility of being able to “tune” the electronic coupling between the chlorophylls and thereby modulate the energy of the chlorophylls-zeaxanthin charge-transfer state. In other words, they should be able to switch the energy-quenching process on or off. “The next step is to examine the energy quenching mechanism in the rest of the Photosystem II complex to see how it is used to regulate the flow of energy throughout the light harvesting system,” said Fleming. Support for this research came from the U.S. Department of Energy's Office of Basic Energy Sciences through its Chemical Sciences, Geosciences, and Biosciences Division. Berkeley Lab is a U.S. Department of Energy national laboratory located in Berkeley, California. It conducts unclassified scientific research and is managed by the University of California. |
|
|
|
|
Related topics - search form: |
|
|