|
Related Topics: |
|
|
|
Current News |
|
Chemistry A to Z |
|
About Internetchemistry |
Ruthenium in a Clinch |
|
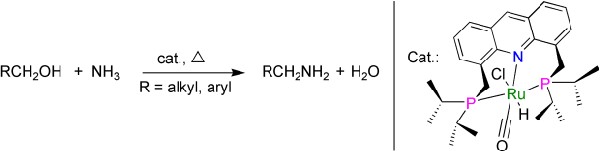
Amines are needed for the production of pharmaceuticals, fine chemicals, agrochemicals, plastics, dyes, pigments, and additives. Most important are compounds with a terminal amino group (–NH2), known as primary amines. However, the selective synthesis of primary amines remains a challenge. David Milstein and Chidambaram Gunanathan at the Weizmann Institute of Science (Israel) have now developed a new catalyst that allows the selective synthesis of primary amines directly from alcohols and ammonia under mild conditions. As they report in the journal Angewandte Chemie, their process involves a complex in which the central ruthenium ion is grasped by a ligand like a pincer. |
|
|
The production of primary amines is so difficult because these products are very reactive and can thus continue to react to form secondary and tertiary amines as undesired byproducts. Previous methods of preparation also mostly require large amounts of toxic reagents. Most large-scale industrial processes are based on the reaction of alcohols with ammonia and require very high temperatures and pressures. This results in a mixture of amines, alkanes, and alkenes as byproducts. A selective catalytic synthesis of primary amines from alcohols and ammonia under mild conditions, without waste products, would be ecologically and economically beneficial.
Credit: Image by Wiley The Israeli scientists have now developed a catalyst that fulfils these wishes: a novel, air-stable ruthenium complex. With two phosphorus-containing “arms”, an organic ligand grasps the ruthenium ion from two sides, holding it like a pincer. A third binding site for the metal ion is provided by a nitrogen atom in the ligand’s central aromatic ring system. By using this pincer complex the researchers were able to selectively synthesize primary amines from alcohols and ammonia under mild conditions; large amounts of toxic compounds are not needed, and harmful waste is avoided. The reactions take place in toluene. Surprisingly, even alcohols immiscible in water react very well without solvents in the presence of water (“on water”). This simple, generally applicable reaction delivers good yields of primary amines, and could represent an interesting alternative not only on the laboratory scale, but also on an industrial scale. In the synthesis of complex natural products and drugs, necessary amino groups could also be incorporated by means of this reaction step directly from the corresponding alcohols. |
|
|
|