|
Related Topics: |
|
|
|
Current News |
|
Chemistry A to Z |
|
About Internetchemistry |
Scientists transform carbon dioxide into methanol |
|
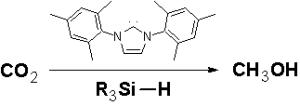
Scientists at Singapore's Institute of Bioengineering and Nanotechnology (IBN) have succeeded in unlocking the potential of carbon dioxide – a common greenhouse gas – by converting it into a more useful product. In the international chemistry journal Angewandte Chemie, the IBN researchers report that by using organocatalysts, they activated carbon dioxide in a mild and non-toxic process to produce methanol, a widely used industrial feedstock and clean-burning biofuel. Organocatalysts are catalysts that are comprised of non-metallic elements found in organic compounds. NHCs such as IMes (1,3-bis-(2,4,6 trimethylphenyl)imidazolylidene) are a form of organocatalysts that are stable and easily stored. They do not contain toxic heavy metals and can be produced easily without high costs. |
|
The scientists made carbon dioxide react by using N-heterocyclic carbenes (NHCs), a novel organocatalyst. In contrast to heavy metal catalysts that contain toxic and unstable components, NHCs are stable, even in the presence of oxygen. Hence, the reaction with NHCs and carbon dioxide can take place under mild conditions in dry air. The IBN scientists showed that only a small amount of NHC is required to induce carbon dioxide activity in a reaction. "NHCs have shown tremendous potential for activating and fixing carbon dioxide. Our work can contribute towards transforming excess carbon dioxide in the environment into useful products such as methanol," said Siti Nurhanna Riduan, IBN Senior Lab Officer, who is also pursuing her Ph.D. under the Scientific Staff Development Award at IBN, one of the research institutes of Singapore's A*STAR (Agency for Science, Technology and Research). Hydrosilane, a combination of silica and hydrogen, is added to the NHC-activated carbon dioxide, and the product of this reaction is transformed into methanol by adding water through hydrolysis. Yugen Zhang, Ph.D., IBN Team Leader and Principal Research Scientist, explained, "Hydrosilane provides hydrogen, which bonds with carbon dioxide in a reduction reaction. This carbon dioxide reduction is efficiently catalyzed by NHCs even at room temperature. Methanol can be easily obtained from the product of the carbon dioxide reaction. Our previous research on NHCs has demonstrated their multiple applications as powerful antioxidants to fight degenerative diseases, and as effective catalysts to transform sugars into an alternative energy source. We have now shown that NHCs can also be applied successfully to the conversion of carbon dioxide into methanol, helping to unleash the potential of this highly abundant gas." Previous attempts to reduce carbon dioxide to more useful products have required more energy input and a much longer reaction time. They also require transition metal catalysts, which are both unstable in oxygen and expensive. Ongoing research at IBN aims to find cheap alternatives for the hydrosilane reagent so that the production of methanol can be even more cost-effective for mass industrial production. "At IBN, we are innovating effective methods of generating clean energy using green chemistry and nanotechnology. In the face of environmental pollution, global warming and increasing demands on diminishing fossil fuel resources, we hope to provide a viable alternative energy option for industry, and effective sequestration and conversion of carbon dioxide," said IBN Executive Director. Jackie Y. Ying, Ph.D. |
||
|
|
||
|
Angewandte Chemie International Edition, press release: |
||
Snatched From the AirCarbon dioxide as a carbon source - a carbene catalyst opens new perspectives It’s the reason why chemists envy green plants: by using photosynthesis, plants can easily fix the carbon dioxide that is so plentiful in air to make biomass, or organic compounds. Chemists would also like to be able to simply produce carbon compounds out of CO2 from air. In contrast to the usual sources of carbon used today - fossil fuels and natural gas - carbon dioxide is a renewable resource and an environmentally friendly chemical reagent. Unfortunately, its carbon–oxygen bonds are too strong to be broken easily. Researchers working with Yugen Zhang and Jackie Y. Ying at the Institute of Bioengineering and Nanotechnology in Singapore have now developed a novel reaction scheme by which CO2 can be efficiently converted into methanol under very mild conditions. As reported in the journal Angewandte Chemie, it is based on an N-heterocyclic carbene catalyst and a silane as the reducing agent. The basic framework of an N-heterocyclic carbene is a five-membered ring made of two nitrogen and three carbon atoms. Instead of having the usual four bonds, one of these carbon atoms only has two. The two electrons left over in the form of a lone pair, which makes this species highly reactive - reactive enough to attack CO2. The researchers in Singapore produced the carbene catalyst used in the reaction in situ from a precursor. The carbene activates the CO2, but is then split off again to end the reaction cycle in its original state. The formal reaction partner is a hydrosilane, an organosilicon compound that acts as a reducing agent. The reaction product into which the CO2 is converted can easily be collected in the form of methanol in the last step of the reaction series. Methanol is an important starting material for many chemical syntheses and serves as an alternative fuel and as a raw material for the production of energy in methanol fuel cells. The big advantage: unlike prior reaction mechanisms using metal-containing catalysts, air can be used as the source of the CO2 because the carbene catalyst is not sensitive to oxygen. The carbene is more efficient than the metal-containing catalysts as well, and the reaction can be carried out under very mild conditions. |
|
|
|